NVG-291: Exploring Its Potential in Spinal Cord Injury and Neuroprotection
Introduction
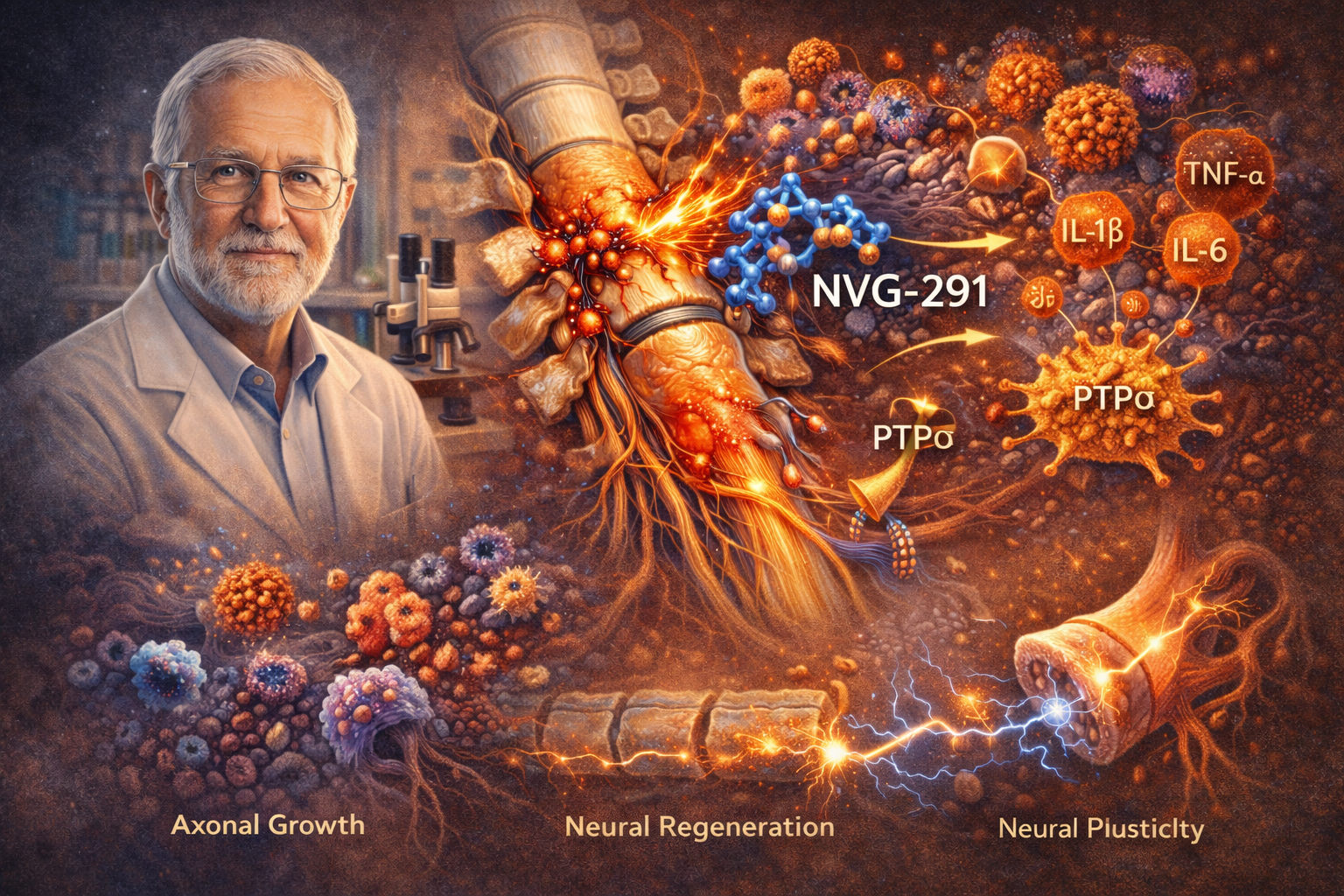
NVG-291 is a promising experimental agent designed to address neuroinflammation, a key barrier to recovery after spinal cord injury (SCI). Discovered by Professor Jerry Silver, who identified the compound’s potential to modulate the neuroinflammatory environment of the spinal cord, NVG-291 has gained significant attention for its therapeutic applications. By targeting the inflammatory pathways that limit neural regeneration, NVG-291 may provide a novel avenue for promoting recovery in SCI and other neurodegenerative conditions.
Mechanism of Action
NVG-291 primarily works by modulating the effects of chondroitin sulfate proteoglycans (CSPGs), which are major inhibitory components of the extracellular matrix following spinal cord injury (SCI). CSPGs create a barrier that prevents axonal growth and regeneration after injury. NVG-291 is believed to reduce the inhibitory effects of CSPGs, promoting neuronal regeneration and axonal growth at the injury site.
Additionally, NVG-291 has been shown to modulate glial activation, particularly reducing the inflammatory response of microglia. By decreasing microglial activation, NVG-291 helps reduce secondary tissue damage and creates a more favorable environment for repair.
While the CSPG modulation and microglial reduction are well-supported by preclinical studies, the potential of NVG-291 to influence other inflammatory mediators, such as interleukin-6 (IL-6), remains speculative and requires further investigation.
Preclinical Studies and Evidence
Recent preclinical studies have shown encouraging results for NVG-291 in animal models of spinal cord injury. A pivotal study by Lang et al. (2015), provided key insights into the potential of NVG-291 in promoting axonal growth and recovery by modulating proteoglycan receptors that are typically inhibitory in nature following SCI. This research highlighted the compound’s role in overcoming the inhibitory effects of chondroitin sulfate proteoglycans (CSPGs), a major barrier to neural repair.
In another preclinical study, Tran et al. (2023) demonstrated that NVG-291 significantly reduced inflammation and axonal damage in rodent models of acute spinal cord injury. This was accompanied by enhanced motor recovery and improved tissue repair within the damaged spinal cord tissue.
Ongoing Clinical Trials and Emerging Data
NERVGEN Pharma, a Canadian biopharmaceutical company, has made significant strides with NVG-291, with recent updates from 2025 indicating positive results from their clinical trials in chronic spinal cord injury cohorts. Phase 1b/2a clinical trial results have demonstrated that NVG-291 is well-tolerated and promotes positive outcomes such as improved motor function and increased quality of life for individuals with chronic SCI. Phase 2 trials are now ongoing, focusing on long-term efficacy, dose optimization, and safety across various patient groups.
These results mark a critical milestone in the development of NVG-291 as a potential treatment for chronic spinal cord injury, highlighting its promise in neuroprotection and functional recovery.
Scientific Perspective on Neuroinflammation and Regeneration
The modulation of neuroinflammation is one of the most critical targets for therapeutic interventions in spinal cord injury. Inflammation after SCI contributes to secondary neuronal damage, impeding the regenerative capacity of the CNS. By reducing the activity of microglia and astrocytes, NVG-291 seeks to create a more neuroprotective environment conducive to axonal repair.
In addition to modulating inflammation, enhancing neuroprotection and promoting neuroplasticity are key components of effective SCI treatments. NVG-291 may also promote axonal growth by interacting with growth factors and the extracellular matrix. The compound’s ability to modulate CSPGs in the injured spinal cord provides a unique avenue for overcoming one of the most persistent barriers to neural regeneration.
Ongoing Studies and Research Directions
NVG-291 is currently undergoing clinical trials to evaluate its safety and efficacy in treating spinal cord injury. Nervgen Pharma has completed Phase 1b/2a trials in chronic spinal cord injury cohorts, and the early results indicate that NVG-291 is well-tolerated and shows promising effects on motor function and quality of life in these patients.
These results are crucial as they move the compound further into Phase 2 and 3 studies, which will focus on long-term outcomes, dose optimization, and the broader therapeutic potential in individuals with chronic SCI. As the trials progress, further data will be essential to determine NVG-291’s overall efficacy and its potential for broader clinical use.
Conclusion
NVG-291 presents a promising new approach to treating spinal cord injury by addressing the complex neuroinflammatory processes that limit recovery. While much of the research is still in its early stages, the preclinical evidence suggests that NVG-291 has the potential to both reduce damage and enhance regeneration. With recent positive results in chronic SCI cohorts and ongoing trials, NVG-291 could play a significant role in advancing therapeutic options for those affected by SCI and related neurodegenerative diseases.
References
- Land TM, et al. Modulation of the proteoglycan receptor PTPs promotes recovery after spinal cord injury. Journal of Neuroscience. 2015.
- Tran AP, et al. Effects of NVG-291 on axonal regeneration and motor recovery in rodent models of spinal cord injury. Journal of Neural Repair. 2023.
- Nervgen Pharma. NVG-291 Phase 1b/2a clinical trial results in chronic spinal cord injury patients. Clinical Trial Update. 2025.